Developing Clinically Relevant Metastatic Colorectal Cancer Models: From Carcinogens to Xenografts
Colorectal cancer (CRC) is common and deadly, making up an estimated 7.9% of all new cancer cases and 8.7% of all cancer deaths in the U.S. last year.1 Cancer mortality often results from metastasis, which for colorectal cancer, happens frequently; one study estimated that 35% of patients present with metastatic disease at the time of diagnosis, and 50% of non-metastatic cases progress to metastatic colorectal cancer (mCRC).2 To improve these clinical outcomes, more effective treatments for metastatic colorectal cancer are needed. Underlying this need is the foundational requirement for more clinically-relevant metastatic colorectal cancer models in preclinical research so that promising therapeutic candidates can be identified and fast-tracked to clinical research.
Figure 1. CRC By the Numbers

- Current standards of care for metastatic colorectal cancer
- Advantages and drawbacks of several preclinical model systems, with a special focus on murine colorectal cancer xenograft models
- Promising results with orthotopic xenograft mouse models for modeling metastatic colorectal cancer
The Current Treatment Landscape for Metastatic Colorectal Cancer
Metastasis from the colon or rectum commonly occurs in the liver, but may also happen in the lungs, bones, brain, or spinal cord.3 Median overall survival (OS) has doubled over the past two decades due to improvements in early detection and treatment of metastatic disease, the efficacy of systemic therapies, biomarker-based patient selection, and promising new data regarding targeted treatments like sotorasib and adagrasib.4 Tragically, once diagnosed with mCRC, median OS is only about thirty months and continued development of more effective treatments is required to improve clinical outcomes and prolong patients’ lives.4
Standard treatment strategy involves combining surgical ablation of metastatic sites paired with systemic drug delivery. Chemotherapy for mCRC typically includes some combination of folinic acid (FOL), 5-fluorouracil (F), oxaliplatin (OX), capecitabine (CAP), or irinotecan (I), together referred to as FOLFOX, CAPOX, FOLFIRI, or CAPIRI.5 Chemotherapy is often combined with angiogenesis inhibitors, such as epidermal growth factor receptor (EGFR) or vascular endothelial growth factor (VEGF) inhibitors.
Other somewhat successful therapies include the PD-1 inhibitors, pembrolizumab and nivolumab, and the BRAF inhibitor, vemurafenib. Unfortunately, many of these therapies run into efficacy problems due to the genetically heterogeneous population of mCRC cancers. Anti-angiogenesis drugs are ineffective in mCRC patients with KRAS mutations, and BRAF inhibitors fail against patients with BRAF mutations, two populations that do not typically overlap.4,6,7 Likewise, BRAF and PD-1 inhibitors are generally only effective against patients with deficient mismatch repair (dMMR) CRC, which is only about 5% of mCRC patients.5
Developing Effective Therapies with Clinically Relevant mCRC Models
In vivo animal models can provide invaluable insights by mimicking clinical characteristics seen in humans, such as disease etiology, progression, metastases, and critically, responses to preclinical drug candidates. However, bridging the physiological gap between common animal models (i.e., mice), humans, and cancer has not been easy. For more than fifty years, researchers have tested and refined many in vivo colorectal cancer models, and in those years, some critical features have been established.
Efficacious mCRC models must:8
- Recapitulate development of invasive carcinoma and metastatic potential
- Reflect inter-individual molecular diversity within cancer
- Mimic the tumor’s physiological microenvironment
Furthermore, to be of use to drug developers, preclinical models need to provide these benefits while minimizing costs and time-to-result, while maximizing throughput.
The Array of Available Mouse Models for mCRC
Figure 2. Mouse Model of Human Colorectal Cancer (Photo Credit: NIH)
Mouse models initiate cancerous growth within the organism via one of three general methods: induction via exposure to carcinogens, genetic engineering, or transplant of cancerous cells or tissue section. And to varying degrees, they meet the clinical relevance, cost, and throughput requirements of drug developers listed above. Let’s take a look at each colorectal cancer model type to get a better sense of the advantages and drawbacks of using each.
Carcinogen-Induced Colorectal Cancer Models (CIMs)
With CIMs, mice are exposed to chemicals that cause tumorigenesis (i.e., the initial formation of a tumor in the body). Exposure can be implemented with direct application to an organ, injection into the peritoneal cavity, or administration with the mouse's diet. Azoxymethane (AOM) and methylazoxymethyl acetate are common carcinogens used for carcinogenesis and are typically administered with intraperitoneal injection. These agents are metabolized to methylazoxyformaldehyde which can alkylate guanine and thymine, and typically induce tumor growth in the small intestine.9,10 Other agents used include N-methyl-N-nitrosourea (MNU), which induces intestinal tumorigenesis via direct rectal application or ingestion.11
Administration via ingestion has served to model dietary influences on the development of colorectal cancer and progression to mCRC. For example, consumption of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP), produced by cooking meat and fish at high temperatures, has been found to induce colitis and adenocarcinomas in mouse models.12,13 Along with diet, CIMs can model the effects of chronic inflammatory bowel disease. Dextran sodium sulfate (DSS), typically administered in drinking water, damages the colonic epithelium, leading to chronic inflammation, and DSS is often combined with other agents (i.e., AOM), to study colitis-induced cancer.8 Finally, different CIMs lead to distinct mutational profiles: the AOM model leads to KRAS mutagenesis, but no APC or P53 mutations, whereas, PhIP and MNU models induce APC mutations, but not P53 or KRAS mutations.8 Overall, CIMs have been useful in the early study of colorectal cancer, yet have little clinical relevance for modeling mCRC.
Advantages:8,14
- Uniquely model dietary exposure to carcinogens and chronic inflammation
- Captures the human progression from crypt foci to adenoma to carcinoma
Disadvantages:8,15,16
- Often induce multiple primary tumors, which does not reflect the typical clinical progression of mCRC in humans
- Models rarely develop to be invasive or metastatic
- Difficult to recapitulate genetic drivers of colorectal cancer
- Tumor development takes considerable time
Genetically-Engineered Colorectal Cancer Mouse Models (GEMMs)
The earliest GEMMs of colorectal cancer created were found using the MNU model described above. The mouse model, known as APCMin, effectively develops intestinal neoplasia, but suffers from poor mortality due to the development of intestinal obstruction with aging, and rarely survives long enough to develop mCRC.17 The combination of APC mutant lines with carcinogenic agents (i.e., AOM), can speed up tumorigenesis to achieve metastasis.18 Likewise, the use of the C57A/J over the C57BL/6 as the background for APCMin increases invasiveness and leads to carcinogenesis within the colon.19
Germline mutations tend to cause tumor growth in off-target organs as well as within the digestive tract, which has limited clinical relevance to human mCRC. Thus, conditional activation of oncogenes or knock-out of tumor suppressors using the Cre-loxP system has been particularly useful, especially when Cre expression is controlled by tissue-specific promoters.8 For example, Villin-Cre mice exhibit Cre expression within the epithelial cells of the whole intestine, whereas Fabpl-Cre mice express Cre in the distal small intestine, cecum, and colon and both have been useful in modeling some aspects of colorectal cancer.
Such methods have led to the generation of many different GEMMs with varying tissue specificity, metastatic potential, and progression. These models have served as a useful platform for studying the role of APC, KRAS, and P53 in colorectal cancer; however, only a handful of the nearly one hundred different GEMMs mimic the clinical characteristics of metastasis.8
Advantages:8,20
- Several models can be used to model metastasis in colorectal cancer
- Reflect details of human mCRC microenvironment
- Includes systemic immune response to cancer
Disadvantages:8,21
- Doesn’t reflect the stepwise genetic or clinical progression of mutations
- Lack of tumor heterogeneity
- Require time-consuming breeding processes
Subcutaneous Colorectal Cancer Xenograft Mouse Models
Subcutaneous engraftment into immunodeficient mouse models has a lot of experimental advantages, including easy implantation (i.e., high take rate and surgical procedure), high throughput, and straightforward tumor growth or drug efficacy monitoring.8 This approach can be done using common cell lines, such as HCT116, which can be genetically manipulated to evaluate the role of specific genes or patient-derived tumor cells or tissue from surgical resection or biopsy, which can be more reflective of real-life human tumors.
Yet, for modeling mCRC, these models have significant drawbacks, especially those that use tumor cells lines. Cell lines are (mostly) genetically homogeneous and bear little genetic or phenotypic resemblance to the tumors they are derived from, due to years of passage.22 They also are missing the interaction between tumor and stroma (i.e. connective tissue, blood vessels, and inflammatory cells that are interspersed between the malignant cells and normal host tissues), a critical piece of the tumor microenvironment. In addition, mCRC models have been developed through intravenous injection of cell suspensions, but these accomplish little relevance to the clinic and don’t mimic any of the early metastatic cascade seen in CRC.23
Another approach that overcomes the issues with using cell lines, is the use of patient-derived tumor samples for subcutaneous xenografts (subcutaneous PDX). The Rostock University Medical Center’s single-center, 11-year study successfully integrated biobanking and tumor model establishment of human colorectal carcinoma (both patient-derived in vitro and matched in vivo models). The Hansestadt Rostock colorectal cancer collection reflects the genetic heterogeneity found in the CRC patient population and provides excellent tools for preclinical research.24 Many studies have also confirmed that these models act as superb models for predicting clinical treatment responses and can be used to direct clinical decisions.25 One study by Bertotti et al. also demonstrated that implantation of liver metastases using a subcutaneous PDX model closely resembled clinical treatment response to cetuximab.26
These results are promising, though there remains some dispute over the utility of this system. Take rates differ between protocols, and there is conflicting data about clinical and molecular correlations with model establishment. Some groups suggest that PDX models are only established with more aggressive tumor types, yet others don’t.8
Advantages:8
- Many studies have used models to recapitulate clinical findings
- Reflects histologic architecture, transcriptional profile, chromosomal instability, and pharmacologic response seen in clinic
- Readily-available validated PDX models with genetic and molecular characteristics found in the clinic
- Easy to measure tumor growth using calipers
Disadvantages:8
- Not an accurate model of the metastatic cascade, though metastatic tumors can be implanted and analyzed subcutaneously
- Microenvironment differs greatly from that of the primary (i.e., colon) or secondary (i.e., liver) tumor
- Human stroma is replaced by mouse components
- Immunodeficient host can’t be used for preclinical studies with immunotherapies
- High cost
- Model establishment can take a long time (~12 months)
- May select for more aggressive tumor types
Orthotopic Colorectal Cancer Xenograft Mouse Models
While the first orthotopic xenograft mouse models used CRC cell lines, many groups have now implemented successful orthotopic PDX (O-PDX) models, and they’ve become popular for their ability to recapitulate the clinical progression of mCRC.
Figure 3. O-PDX models capture many characteristics of tumors in the clinic and combine it with the physiological relevance of orthotopic (which means “the correct place”) implantation.

The most popular implantation site is the cecum, and it’s frequently used, though there are some critiques about the methodology and other protocols that involve subserosal or submucosal implantation have been developed.27,28 While O-PDX does require a high degree of surgical skill for implantation, it has been shown to capture the invasiveness, critical tumor microenvironmental features such as morphology, genetic heterogeneity, and transcriptional profile, and robust ability to model metastasis and end stages of mCRC in a short period of time.29-33 The ability to model metastasis is a major advantage of O-PDX over subcutaneous PDX models and several studies had shown that O-PDX models also mimic pharmacological responses seen in the clinic.8,32,33 Transduction of PDX samples with reporter genes such as GFP or luciferase, has further facilitated the preclinical and clinical utility of O-PDX, as tracking tumor growth or metastasis is often challenging with these models.
Despite these promising results, cost and low tumor take rate have significantly limited the more widespread adoption of O-PDX for large-scale preclinical screening and “co-clinical” trials.
Advantages:8,32,33
- Accurate model for mimicking early- and late-stage metastatic cascade
- Metastatic tumors mimic pharmacological responses seen in the clinic
- Metastatic tracking can be done by transducing PDX with reporter genes
- Reflects histologic architecture, transcriptional profile, chromosomal instability, and pharmacologic response seen in clinic
- Tumor microenvironment is similar to that found in the patient
- Reflect details of human mCRC microenvironment (i.e. stromal components)
- Readily-available validated PDX models with genetic and molecular characteristics found in the clinic
Disadvantages:8,32,33
- Human stroma is replaced by mouse components
- Immunodeficient host can’t be used for preclinical studies with immunotherapies
- High cost
- Model establishment can take a long time (~12 months)
- May select for more aggressive tumor types
Access Custom PDX Models of mCRC with Certis
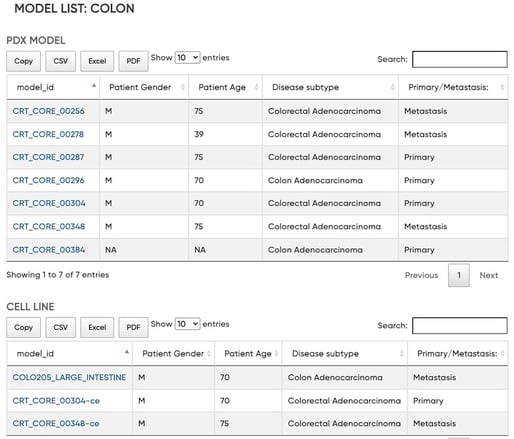
At Certis, we’re committed to improving the translation of mCRC therapies by facilitating clinically accurate preclinical research through the use of PDX mouse models. We’re continually adding to our well-characterized bank of CRC tumors that include primary and metastatic colorectal adenocarcinoma models, some with matched cell lines, that capture the breadth and heterogeneity found in colorectal cancer.
Figure 4. Certis Colorectal Cancer PDX Models and Matched Cell Lines
By outsourcing PDX model development and other preclinical assays to us, we can reduce your time-to-result and provide access to actionable data, enabling you to make better preclinical decisions. Register for access to our searchable tumor database now. For more information, email us at busdev@certisoncology.com.
References
- Cancer Stat Facts: Colorectal Cancer. SEER website. Accessed March 10, 2022.
- Zacharakis M, Xynos ID, Lazaris A, et al. Predictors of Survival in Stage IV Metastatic Colorectal Cancer. Anticancer Res. 2010;30(2):653-660.
- Metastatic Colorectal Cancer. Cancer Treatment Centers of America website. Sept. 21, 2021. Accessed Feb. 28, 2022.
- Cutsem EV, Cervantes A, Adam R, et al. ESMO Consensus Guidelines for the Management of Patients with Metastatic Colorectal Cancer. Ann Oncol. 2016;27(8):1386-1422.
- Wrobel P, Ahmed S. Current Status of Immunotherapy in Metastatic Colorectal Cancer. Int J Colorectal Dis. 2019;34(1):13-25.
- Kopetz S, Desai J, Chan E, et al. Phase II Pilot Study of Vemurafenib in Patients With Metastatic BRAF-Mutated Colorectal Cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2015;33(34):4032-4038.
- Kafatos G, Niepel D, Lowe K, et al. RAS Mutation Prevalence Among Patients with Metastatic Colorectal Cancer: A Meta-Analysis of Real-World Data. Biomark Med. 2017;11(9):751-760.
- Bürtin F, Mullins CS, Linnebacher M. Mouse Models of Colorectal Cancer: Past, Present and Future Perspectives. World J Gastroenterol. 2020;26(13):1394-1426.
- Papanikolaou A, Shank RC, Delker DA, Povey A, Cooper DP, Rosenberg DW. Initial Levels of Azoxymethane-Induced DNA Methyl Adducts are Not Predictive of Tumor Susceptibility in Inbred Mice. Toxicol Appl Pharmacol. 1998;150(1):196-203.
- Rosenberg DW, Giardina C, Tanaka T. Mouse Models for the Study of Colon Carcinogenesis. Carcinogenesis. 2009;30(2):183-196.
- Sugimura T, Terada M. Experimental Chemical Carcinogenesis in the Stomach and Colon. Jpn J Clin Oncol. 1998;28(3):163-167.
- Chen JX, Wang H, Liu A, Zhang L, Reuhl K, Yang CS. From the Cover: PhIP/DSS-Induced Colon Carcinogenesis in CYP1A-Humanized Mice and the Possible Role of Lgr5+ Stem Cells. Toxicol Sci Off J Soc Toxicol. 2017;155(1):224-233.
- Nakagama H, Ochiai M, Ubagai T, et al. A Rat Colon Cancer Model Induced by 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine, PhIP. Mutat Res. 2002;506-507:137-144.
- De Robertis M, Massi E, Poeta ML, et al. The AOM/DSS Murine Model for the Study of Colon Carcinogenesis: From Pathways to Diagnosis and Therapy Studies. J Carcinog. 2011;10:9.
-
Nambiar PR, Girnun G, Lillo NA, Guda K, Whiteley HE, Rosenberg DW. Preliminary Analysis of Azoxymethane Induced Colon Tumors in Inbred Mice Commonly Used as Transgenic/Knockout Progenitors. Int J Oncol. 2003;22(1):145-150.
- Papanikolaou A, Wang QS, Papanikolaou D, Whiteley HE, Rosenberg DW. Sequential and Morphological Analyses of Aberrant Crypt Foci Formation in Mice of Differing Susceptibility to Azoxymethane-Induced Colon Carcinogenesis. Carcinogenesis. 2000;21(8):1567-1572.
- Shoemaker AR, Gould KA, Luongo C, Moser AR, Dove WF. Studies of Neoplasia in the Min Mouse. Biochim Biophys Acta. 1997;1332(2):F25-48.
- Tanaka T, Suzuki R, Kohno H, Sugie S, Takahashi M, Wakabayashi K. Colonic Adenocarcinomas Rapidly Induced by the Combined Treatment with 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine and dextran sodium sulfate in Male ICR Mice Possess Beta-Catenin Gene Mutations and Increases Immunoreactivity for Beta-Catenin, cyclooxygenase-2 and inducible nitric oxide synthase. Carcinogenesis. 2005;26(1):229-238.
- Sødring M, Gunnes G, Paulsen JE. Spontaneous Initiation, Promotion and Progression of Colorectal Cancer in the Novel A/J Min/+ Mouse. Int J Cancer. 2016;138(8):1936-1946.
- DuPage M, Jacks T. Genetically Engineered Mouse Models of Cancer Reveal New Insights about the Antitumor Immune Response. Curr Opin Immunol. 2013;25(2):192-199.
- Huang D, Sun W, Zhou Y, et al. Mutations of Key Driver Genes in Colorectal Cancer Progression and Metastasis. Cancer Metastasis Rev. 2018;37(1):173-187.
- Brattain MG, Fine WD, Khaled FM, Thompson J, Brattain DE. Heterogeneity of Malignant Cells from a Human Colonic Carcinoma. Cancer Res. 1981;41(5):1751-1756.
- Murdocca M, Capuano R, Pucci S, et al. Targeting LOX-1 Inhibits Colorectal Cancer Metastasis in an Animal Model. Front Oncol. 2019;9:927.
- Mullins CS, Micheel B, Matschos S, et al. Integrated Biobanking and Tumor Model Establishment of Human Colorectal Carcinoma Provides Excellent Tools for Preclinical Research. Cancers (Basel). 2019;11(10):1520.
- Hidalgo M, Bruckheimer E, Rajeshkumar NV, et al. A Pilot Clinical Study of Treatment Guided by Personalized Tumorgrafts in Patients with Advanced Cancer. Mol Cancer Ther. 2011;10(8):1311-1316.
- Bertotti A, Migliardi G, Galimi F, et al. A Molecularly Annotated Platform of Patient-Derived Xenografts ("Xenopatients") Identifies HER2 as an Effective Therapeutic Target in Cetuximab-Resistant Colorectal Cancer. Cancer Discov. 2011;1(6):508-523.
- Yang JL, Seetoo Dq, Wang Y, et al. Urokinase-Type Plasminogen Activator and its Receptor in Colorectal Cancer: Independent Prognostic Factors of Metastasis and Cancer-Specific Survival and Potential Therapeutic Targets. Int J Cancer. 2000;89(5):431-439.
- Céspedes MV, Espina C, García-Cabezas MA, et al. Orthotopic Microinjection of Human Colon Cancer Cells in Nude Mice Induces Tumor Foci in All Clinically Relevant Metastatic Sites. Am J Pathol. 2007;170(3):1077-1085.
- Lee WY, Hong HK, Ham SK, Kim CI, Cho YB. Comparison of Colorectal Cancer in Differentially Established Liver Metastasis Models. Anticancer Res. 2014;34(7):3321-3328.
- Visioli A, Giani F, Trivieri N, et al. Stemness Underpinning All Steps of Human Colorectal Cancer Defines the Core of Effective Therapeutic Strategies. EBioMedicine. 2019;44:346-360.
- Julien S, Merino-Trigo A, Lacroix L, et al. Characterization of a Large Panel of Patient-Derived Tumor Xenografts Representing the Clinical Heterogeneity of Human Colorectal Cancer. Clin Cancer Res Off J Am Assoc Cancer Res. 2012;18(19):5314-5328.
- Sebolt-Leopold JS. Development of Preclinical Models to Understand and Treat Colorectal Cancer. Clin Colon Rectal Surg. 2018;31(3):199-204.
- Heijstek MW, Kranenburg O, Borel Rinkes IH. Mouse Models of Colorectal Cancer and Liver Metastases. Dig Surg. 2005;22(1-2):16-25.
About the Author
Elie Diner has a PhD in bioengineering and 12 years of research experience in microbiology, synthetic biology and immunology. During his time at the bench, he developed a passion for effective science communication and eventually transitioned into a career as a professional science and content writer. He's authored 12 peer-reviewed scientific publications and numerous blogs, whitepapers, and eBooks for life science companies.
Back to Feed