How to Overcome the Translation Gap in Cancer Research
Custom Solutions, Better Translation
The ultimate goal of drug development in oncology is to identify drug candidates that will successfully advance through the clinical trial pipeline to FDA approval, thus offering individuals with cancer the hope of better, longer lives.
However, there is a translation gap between preclinical discovery and clinical success: very few preclinical drug candidates that looked promising in the early stages of drug development survive the path to FDA approval.
The likelihood of overall drug approvals from phase I clinical trials hovers around 10%.1-3
From Gap to Chasm: Translation Challenges in Oncology
In oncology, where drug development activity has reached a fever pitch, the gap has grown into a deep and persistent chasm. Over 95% of oncology drugs that enter the clinic fail to receive FDA approval as drug developers clammer to identify the next blockbuster therapeutic that will transform the current standard of care.3,4
Consequently, massive amounts of time and money – and the time and well-being of altruistic clinical trial volunteers and individuals with cancer – are being invested at each stage of the pipeline, with very few new transformative drugs to show for it at the end.
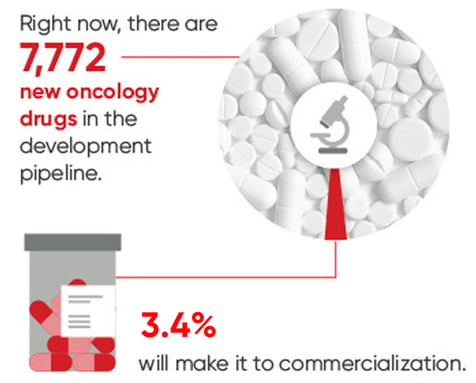 Figure 1. Wong CH et al. Biostatistics. (2019).
Figure 1. Wong CH et al. Biostatistics. (2019).
Personalized Medicine and Targeted Cancer Therapeutics
While classic chemotherapeutics remain widely used across many cancer types, the industry has moved mainly towards developing personalized oncology drugs targeted to a specific genetic or protein biomarker identified. This approach has helped circumvent chemotherapeutics' unpleasant, systemic side effects and addresses the heterogenous nature of cancer.
Currently, targeted therapies use new modalities for cancer treatment, including cell and gene therapies, therapeutic antibodies, vaccines, and microbes. Several of these therapies have been successful on their path to market, yet, despite their targeted nature, many face similar translation challenges as untargeted drugs.
The Translation Gap is a Preclinical Model Gap
The irrelevance of preclinical experiments and models to the clinical context of human cancers has been blamed for the current translation chasm.5 A preclinical model that does not recapitulate the clinical characteristics of the disease may not be able to predict crucial human-relevant safety, pharmacological, and mechanistic information before a candidate drug moves into clinical testing.
For example, two-dimensional (2D) cultures with cancer cell lines have been a foundational and widely used high-throughput tool for drug screening. However, 2D culture does not mimic important in vivo cell characteristics, such as cell-cell or cell-matrix interactions. In addition, the tumor microenvironment is a heterogenous amalgamation of tumor and normal cells, a far cry from the nearly pure monoculture of immortalized cells found in a petri dish. Furthermore, complex tumor behaviors such as metastasis are impossible to simulate in a 2D environment, making these models very limiting and difficult to predict clinical relevance.
Three-dimensional (3D) cell culture systems are better models of physical tumor structure than 2D systems, but they do not mimic in vivo elements like vasculature, microenvironment, or human immune response. Genetically-engineered mouse models (GEMMs) are powerful basic science models: Many models are suitable for developing hypotheses and examining the biology of known mutations. However, GEMMs are not ideal tools for drug screening because there can be species-specific pharmacological and safety responses.6
.jpg?width=1053&height=204&name=CMD_Article%20Image%20CvA%20(1).jpg)
Given the ubiquity of the translation gap, there are countless examples to prove this point. Take, for instance, the prototypical case of TGN1412, a CD28 superagonist antibody: When administered to six healthy volunteers at a dose 500-times lower than those found safe in preclinical mouse models, yet induced multisystem organ failure and cytokine storm.7,8
Beyond the One-Size-Fits-All Preclinical Approaches: The Need for Custom Models
Rather than accepting the translation gap as static and unbridgeable, cancer researchers are rethinking how to perform preclinical research under more clinically-relevant conditions. Overcoming the translation gap means developing customized preclinical oncology tools, taking a non-linear path to preclinical vetting, types of studies performed, and mirroring preclinical and clinical endpoints.
Developing Customized, Clinically Relevant Models
Large-scale, high-throughput screening with advanced cell-based technologies has value in a preclinical strategy.5 But in response to the complexities of cancer and human clinical relevance, therapeutics developers should consider expanding their preclinical efforts to include more customized, clinically-relevant approaches.
To meet the demand for models that match the complexity and heterogeneity of cancer, models that use patient-derived tissue have emerged, including 3D organoids, 3D spheroids, and xenografts.5 The benefit of this approach, particularly with patient-derived xenografts (PDX) is that drug developers can mimic complex clinical characteristics, such as metastasis, treatment resistance, and human immune responses. One of the downsides to PDX models is that they are immunocompromised and, thus, not appropriate for testing immuno-oncology drugs. To address this issue, humanized mice that can be used with PDX models have been developed, enabling pharmacological testing, closer interrogation of the human tumor and immune system, and a better way to avoid species-specific effects.9
 Figure 2. Adapted from Akter F et al. Biochim Biophys Acta Rev Cancer. (2021).
Figure 2. Adapted from Akter F et al. Biochim Biophys Acta Rev Cancer. (2021).
In addition, with the vast availability of public and private biorepositories, patient-derived tissue with a broad range of biomarkers and detailed molecular, cellular, histological, and medical metadata is available for custom model creation that mimics real-world clinical cases.10 Once developed, these models can be used for efficacy and safety testing to determine which patient populations benefit most from treatment, leading to a more precise clinical picture for better patient stratification and clinical study planning. Custom model development also facilitates the introduction of markers, such as luciferase, to make tracking tumor growth and metastasis more efficient.
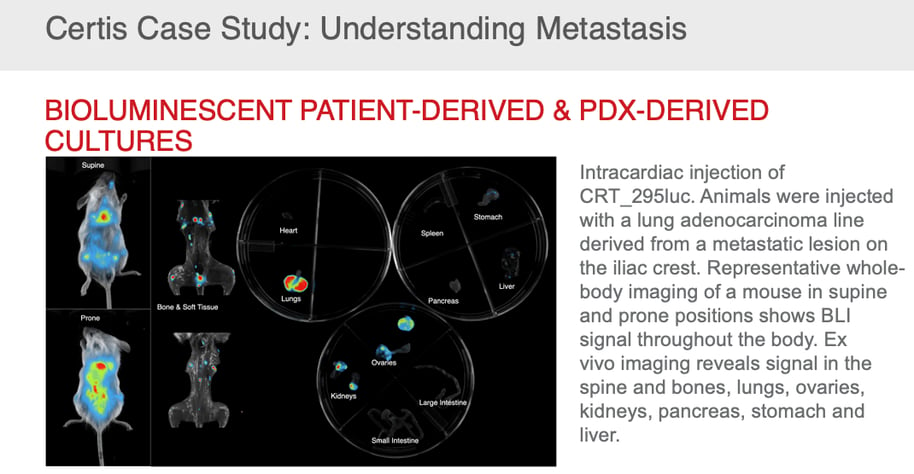
This customized approach has proven successful across several different cancer types, including successful for rare, high-grade cancers. PDX models of high-risk pediatric neuroblastomas, some of which were from relapsed and refractory disease, have been successfully developed and deployed for drug testing on preclinical and clinical timelines.11
Taking an Iterative, Cyclical Approach in Preclinical Cancer Research
The current spectrum of preclinical models offers different degrees of biological accuracy and clinical predictability.
On the low side of predictability are 2D in vitro cell culture models, which offer a high-throughput way to screen for safety and efficacy, yet are essentially blind to the human body's complexities. On the other end of the spectrum are well-characterized PDX models, orthotopically (i.e., in the normal or usual place in the body) implanted into humanized animal models. These models provide a preclinical system as close to the clinical context as possible.
Neither 2D cell cultures nor PDX preclinical models should be used in a vacuum, and results from each system should inform the safety and efficacy studies conducted with the others. Early drug development is often depicted as a linear path, yet an iterative approach to preclinical testing is more appropriate. Results from in vivo animal studies, successful treatment combinations and relevant biomarker studies should inform subsequent preclinical testing (or re-testing) using in vitro models or custom in vivo models that can help drill down on critical mechanistic questions and provide a more accurate preview of what drug developers may find in the clinic.

This iterative approach can even extend into early clinical testing. One example where there is iterative crosstalk between preclinical models and clinical testing is co-clinical trials, where drug testing in humans and mice occurs in parallel.
Co-clinical trials allow cross-talk between a preclinical model and clinical study in real-time, facilitating more accurate patient stratification based on biomarkers and providing insight into drug resistance mechanisms. Ultimately, it provides a way to predict and monitor the safety and efficacy of treatments using personalized mouse avatars.
Numerous trials are testing the effectiveness of the co-clinical strategy across rare and recurrent cancer types. One recent publication used patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck to test the efficacy of combinations of buparlisib and cetuximab on personalized PDX models.1,2 Results from PDX models were used to, in near real-time, revise clinical trials to treat patients with a drug combination that has the highest likelihood of being safe and effective.
Matching Preclinical and Clinical Treatment Regimens and ENdpoints
In many oncology clinical trials, endpoints include overall survival (OS), progression-free survival (PFS), time to progression, and overall response rate. However, preclinical testing typically doesn’t rely on outcomes.13 Rather, they measure growth inhibition, tumor growth delay, or tumor volume change.
The difference in endpoints between preclinical and clinical work can further widen the translation gap. Vismodegib provides a key example: The drug inhibited tumor growth in a preclinical colorectal cancer xenograft model yet showed no clinical benefit in a phase II trial.1,3
Currently, PDX studies lack clear outcome definitions.1,4 The scientific community should consider selecting well-characterized and validated preclinical endpoints that are closely correlated with clinical endpoints.
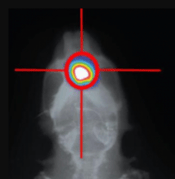
Furthermore, treatment regimens that match those used in the clinic should be recapitulated in preclinical studies. In this respect, custom models of drug resistance or radiosensitivity can be developed by pre-treating models with the respective treatment in vivo. Whole-body and focal radiation (Figure 3 on right) can be used in both in vivo and in vitro studies to mimic clinical scenarios. With the X-Rad 320 radiator, a homogeneous beam delivers a precise radiation dosage to both cells and whole animals enabling Certis scientists to replicate radiation therapy in a laboratory setting.
Leaping The Translation Gap With custom model development
Customized preclinical models are now at the center of successful translation into clinical development and commercialization. As a leader in developing customized oncology models and solutions, Certis Oncology Solutions can bring greater certainty to go/no-go development decisions with clinically relevant, well-characterized, and annotated PDX mouse models.
Custom Models Start with the RIght Tissue
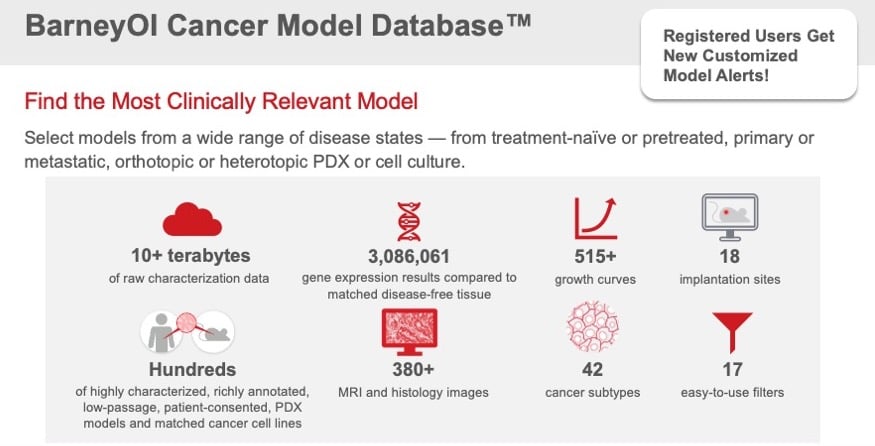
Certis is a life science technology company specializing in custom cancer model development, from advanced cellular and in vivo models of recurrence, resistance, and metastasis, to rare and humanized mouse models. Select from highly characterized, richly annotated models within the BarneyOI Cancer Model Database™, commercially available tissue or cell lines, or client-supplied tumors. Certis also offers standard-of-care treatments, including radiation therapy, to mimic real-world clinical scenarios. Combining its deep scientific expertise with a highly collaborative approach offers therapeutics developers powerful options for leaping the translational gap.
It's Never too Early To Start Thinking About Your Next PReclinical Study
Contact a Certis Business Development Team member now to discuss your unique needs with our custom-developed cancer models.
 Schedule Meeting
Schedule Meeting Email: busdev@certisoncology.com
Email: busdev@certisoncology.com  Call: (858) 988-1033
Call: (858) 988-1033
References
-
Takebe T, Imai R, Ono S. The Current Status of Drug Discovery and Development as Originated in United States Academia: The Influence of Industrial and Academic Collaboration on Drug Discovery and Development. Clin Transl Sci. (2018) 11, 597-606.
-
Dowden H and Munro J. Trends in Clinical Success Rates and Therapeutic Focus. Nat Rev Drug Disc. (2019) 18, 495-496.
-
Schueler J et al. How to Build a Tumor: An Industry Perspective. Drug Discovery Today. Oct. 2022;27(10).
-
Wong CH, Siah KW, Lo AW. Estimation of Clinical Trial Success Rates and Related Parameters. Biostatistics. 2019 Apr 1;20(2):273-286.
-
Foglizzo V, Cocco E, Marchio S. Advanced Cellular Models for Preclinical Drug Testing: From 2D Cultures to Organ-on-a-Chip Technology. Cancers (2022) 14, 3692.
-
Uhl E and Warner N. Mouse Models as Predictors of Human Responses: Evolutionary Medicine. Curr Pathobiol Rep. (2015) 3, 219-223.
-
Attarwala H. TGN1412: From Discovery to Disaster. J Young Pharm. (2010) 2(3):332-336.
-
Suntharalingam G, Perry MR, Ward S, et al. Cytokine Storm in a Phase 1 Trial of the Anti-CD28 Monoclonal Antibody TGN1412. N Engl J Med. (2006) 355(10):1018-1028.
-
Walsh NC, Kenney LL, Jangalwe S, et al. Humanized Mouse Models of Clinical Disease. Annu Rev Pathol. (2017) 12:187-215.
-
Dudová Z, Conte N, Mason J, et al. The EurOPDX Data Portal: An Open Platform for Patient-Derived Cancer Xenograft Data Sharing and Visualization. BMC Genomics. (2022) 23(1):156.
-
Kamili A, Gifford AJ, Li N, et al. Accelerating Development of High-Risk Neuroblastoma Patient-Derived Xenograft Models for Preclinical Testing and Personalised Therapy. Br J Cancer. (2020) 122(5):680-691.
-
Kim HR, Kang HN, Yun MR, et al. Mouse-Human Co-Clinical Trials Demonstrate Superior Anti-Tumour Effects of Buparlisib (BKM120) and Cetuximab Combination in Squamous Cell Carcinoma of Head and Neck. Br J Cancer. (2020) 123(12):1720-1729. doi:10.1038/s41416-020-01074-2
-
Ireson CR, Alavijeh MS, Palmer AM, Fowler ER, Jones HJ. The Role of Mouse Tumour Models in the Discovery and Development of Anticancer Drugs. Br J Cancer. (2019) 121(2):101-108.
-
Collins AT, Lang SH. A Systematic Review of the Validity of Patient Derived Xenograft (PDX) Models: The Implications for Translational Research and Personalised Medicine. PeerJ. (2018) 6:e5981.
ABOUT THE AUTHOR
Elie Diner has a PhD in bioengineering and 12 years of research experience in microbiology, synthetic biology and immunology. During his time at the bench, he developed a passion for effective science communication and eventually transitioned into a career as a professional science and content writer. He's authored 12 peer-reviewed scientific publications and numerous blogs, whitepapers, and eBooks for life science companies.
Back to Feed
